Blog
This is a subtitle for your new post

Trading within the UK and between the UK and Europe just got a lot more complicated
Despite the trade deal between the UK and the EU being hailed as a major success for both sides, the main benefits of the agreement focus on the elimination of trading tariffs and the equalisation of customs rules. So far as the single market (and hence the rules for CE marking) are concerned, the UK is now split up into two parts: Northern Ireland remains within the single market while Great Britain (i.e. Scotland, Wales and England) leaves the single market and is free to set its own rules.
This is what we know so far:
- From 1 January 2021, the UKCA mark will start to replace the CE mark for goods sold within Great Britain.
- The CE mark will continue to be required for goods sold in Northern Ireland.
- Units already in the supply chain at the time the new rules come into effect can continue to be sold under the old rules.
- For most CE marked goods there will be a one year transition period commencing on 1 January 2021, during which time the CE marking will continue to be acceptable as an alternative to the UKCA mark for goods sold in Great Britain.
- Some goods (e.g. certain medical devices) will have an extended transition period up to 2023.
- For all other goods, the UKCA mark will become mandatory at the end of 2021.
- Products will be able to display both the UKCA mark and the CE logo unless and until the rules for those products diverge between the UK and EU.
- It's not expected that any divergence will happen in the near future.
- Products which require third party (i.e Notified Body) involvement as part of their CE marking process will have to use a UK Approved Body instead of (or as well as) an EU Notified Body - see below for more details.
- Manufacturers based within Great Britain will need to identify a representative based within the EU whose name and address can go on the product as a contact point for the Market Surveillance Authorities.
- Manufacturers based outside the UK and selling in the UK will need a UK based representative.
The UKCA Mark
The UKCA mark is the replacement for CE marking in Great Britain. Applying the UKCA mark is exactly like using the CE logo. The Regulations and Directives which created the legal structure for CE marking have now been adopted into UK law and updated to change the terminology and to bring them completely within the control of the UK government.
The following table summarises the terminology equivalence:
New To apply the UKCA mark, manufacturers have to do exactly the same things as they did to apply the CE mark, and in most cases they can use the same standards, risk assessments, test data, and technical files so the only thing which changes is the logo on the product and the words on the Declaration of Conformity.
Products being sold in the EU as well as Great Britain will continue to need the CE logo, so it's likely that most products will have both the UKCA mark and the CE logo on them.
Standards
Those familiar with CE marking will know that the key to the process is the correct application of standards to provide detailed design guidance and recommended solutions for complying with the safety requirements of the CE Directives and Regulations.
Standards will continue to form the bedrock of the product safety landscape in the UK after Brexit. The UK Government will publish lists of 'designated standards' which can be used as the basis of conformity with the UKCA requirements, in exactly the same way as harmonised standards provide a presumption of conformity for CE marking.
Standards will continue to be published in the UK by the British Standards Institute. BSI remains a member of CEN and CENELEC until at least the end of 2021, but only as an EEA member, not as a full EU member. This means BSI's influence on the drafting of harmonised standards is diminished.
It's not currently clear what happens to the UK's obligation to withdraw conflicting national standards and replace them with harmonised standards once BSI is no longer a full member of CEN or CENELEC. The obligation is part of the CEN/CENELEC agreement which national standards bodies must sign up to if they are to become full members and it's probably the most significant factor in the harmonisation of products for sale across the single market. However, it's difficult to see how the commitment to withdraw conflicting national standards is compatible with the full national sovereignty aspect of Brexit. The new UK-EU agreement includes words which indicate that the UK will focus on standardisation at an 'international' level but quite what that means in practice remains to be seen.
Notified Bodies, Approved Bodies
Notified Bodies have an important role to play in the CE marking of many products. The Notified Body may be involved in type testing the design or in ensuring the consistency of production (quality control), or both. Where the Notified Body is involved in checking the conformity of production, their four figure number must accompany the CE logo on the product.
Notified Bodies must be located in an EU Member State, or in a territory where there is a mutual recognition agreement (MRA) between that territory's government and the EU. There is (currently, at least) no such MRA between the UK and the EU, so all Notified Bodies based in the UK ceased to be notified on 31 December 2020.
Most UK based Notified Bodies have now opened offices in European states and have had these appointed as EU Notified Bodies by the EU country where they are based so they can continue to provide Notified Body services in the CE marking field to their clients.
The UK Government has now appointed all the UK based Notified Bodies who requested transfer as UK Approved Bodies. They retain their four figure EU Notified Body number which must go on products alongside the UKCA mark if required by the relevant regulations for that type of product. Approved Bodies can only approve products which are UKCA marked and intended for sale within Great Britain, and EU Notified Bodies can only approve products for CE marking, so a manufacturer who wishes to sell both in the EU and in the UK must appoint both a UK Approved Body and an EU Notified Body. In practice, in most cases this can be the same organisation so long as it has a commercial presence and accreditation in the UK as well as Europe.
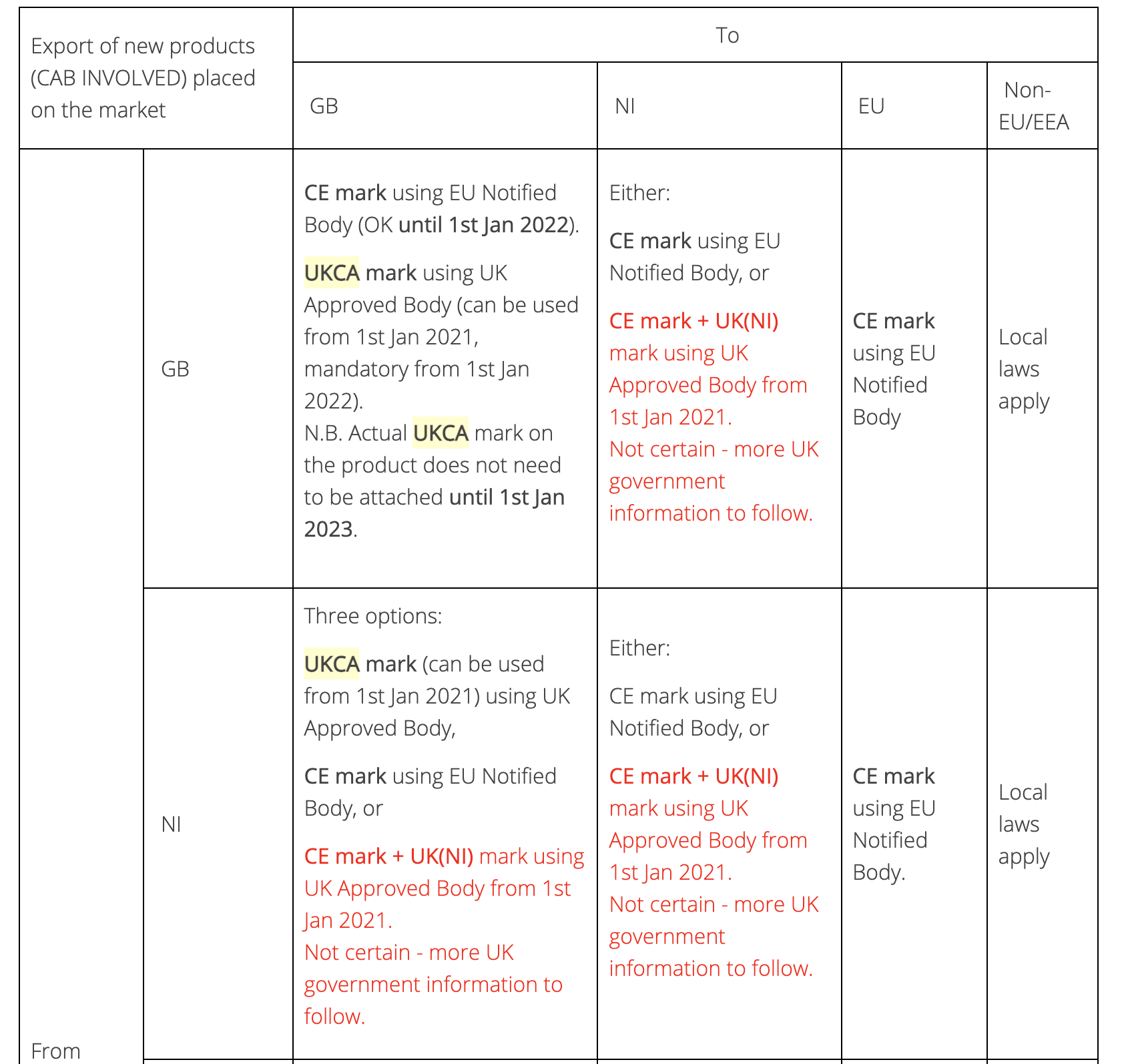
The situation in Northern Ireland is even more complicated because the UK government has guaranteed equivalence between the Northern Irish market and the rest of the UK, even though the CE mark remains mandatory in Northern Ireland. Because a UK Approved Body cannot normally approve goods for CE marking, manufacturers based in Northern Ireland would need to engage the services of a Notified Body from elsewhere in Europe. This would put manufacturers based in Northern Ireland at a disadvantage in comparison with their GB based competitors, so the Northern Ireland Protocol permits a manufacturer based in Northern Ireland to CE mark their products on the basis of certification from a UK Approved Body, but the CE mark on such products must be accompanied by a new logo - the 'UKNI' marking. Products bearing the UKNI mark can be freely traded within the UK but they cannot be sold in the EU, despite them carrying the CE logo.
The following table summarises the situation, and our UKCA FAQ also has more information.
New Authorised Representatives
The UK is no longer part of the EU and the transitional arrangements have expired. This means that any requirement in the CE marking legislation which mandates a presence within the EU can no longer be fulfilled by a UK based office.
Such requirements include the need to identify an Authorised Representative for medical devices, the requirement for an address within Europe for the 'person authorised to compile the technical file' for machinery and the need to identify an EU importer for most of the other CE marking directives and regulations.
This means that manufacturers based outside the EU will need to come to an arrangement with someone who can fulfil this obligation from 1 January 2021. Furthermore, manufacturers based outside the UK who wish to sell products into the UK market will also need a representative within the UK. This is covered in much more detail on our Authorised Representative page, along with further information about new obligations under the Market Surveillance Regulation that come into force in July 2021.
In summary...
Here are the most important points to take away:
- Don't panic! For most products manufacturers still have until the end of 2021 to get themselves organised to apply the UKCA mark.
- For the moment, the technical requirements for the UK and EU markets remain identical, it is only some of the administrative paperwork which changes.
- Manufacturers selling within the EU will need to ensure they have representation in the EU, and manufacturers selling within the UK must have representation within the UK.
As ever, Conformance is here to help you get to grips with these changes. Contact us if you need help.
Please click here to see the original source of the article.